-
MEDICATIONS
- ANALGESICS
- ALLERGY
- ANTIFUNGAL
- ANTIMICROBIAL
- ANTIPARASITICS
- ANTISEPTICS
- BONES AND JOINTS
- COUGH AND ANTIVIRAL
- DERMATOLOGY
- CARDIOLOGY
- GENITOURINARY SYSTEM
- GYNECOLOGY
- HOMEOPATHY
- HAEMORRHOIDS
- ONCOLOGY
- INCREASE POTENCY
- RESPIRATORY SYSTEM
- HORMONES
- IMMUNE SYSTEM
- NEUROLOGY
- ANXIETY AND INSOMNIA
- OPHTHALMOLOGY
- OTHER
- STOMACH. INTESTINES. LIVER
- TOPICAL MEDICATIONS
- WEIGHT LOSS. METABOLIC DISORDERS
-
SUPPLEMENTS
- ANTIOXIDANT COMPLEXES
- BEER YEAST
- CALCIUM, D3, FISH OIL
- EYE VITAMINS
- HERBALS
- IRON PREPARATIONS
- MACRO AND TRACE ELEMENTS
- MONO-AND MULTIVITAMINS
- RESTORATIVE
- SUPPLEMENTS FOR THE DIGESTIVE TRACT, LIVER, KIDNEYS
- TONIC
- VITAMIN AND MINERAL COMPLEXES
- VITAMIN C
- VITAMIN D
- VITAMIN E
- VITAMINS FOR CHILDREN
- VITAMINS FOR MEN
- VITAMINS FOR PREGNANT AND LACTATING
- VITAMINS FOR SKIN, HAIR, NAILS
- VITAMINS FOR WOMEN
- VITAMINS PREMIUM CLASS SOLGAR (SOLGAR)
-
MEDICAL SUPPLIES
-
BEAUTY
-
COSMETIC BRANDS
- BEAUTY CARE (BISI)
- BIODERMA
- BIORGA
- CERAVE (CERAW)
- ZINOVIT
- DNC (DNC COSMETICS)
- DRY DRY (DRY DRY)
- DUCRAY (DUCRE)
- ELANCYL (ELANSIL)
- ELFAPHARM (ELFA PHARM)
- EVA (EVA)
- FILORGA (FILORGA)
- FITO COSMETICS (PHYTO COSMETIC)
- FLORESAN
- HORMETA (ORMETA)
- INSTITUT ESTHEDERM (ESTEDERM)
- IQ BEAUTY (IQ BEAUTY)
- KLORANE (CLORAN)
- KNEIPP (KNIPP)
- KORFF (KORFF)
- LA ROCHE-POSAY (LA ROSH POSE)
- LAINO (LENO)
- LIBREDERM (LIBRIDERM)
- LIERAC
- FILLERS
-
COSMETIC BRANDS
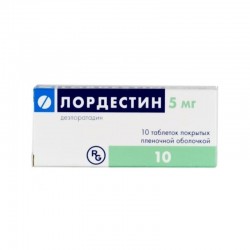
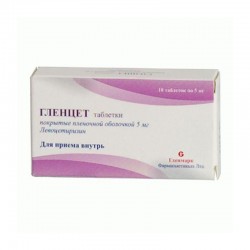
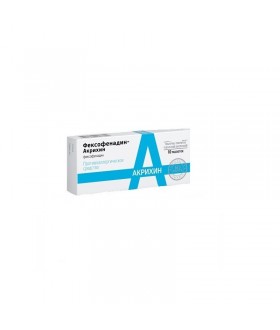
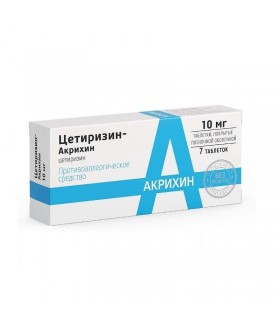
ALLERGY
There are 87 products.
Showing 1-12 of 87 item(s)
Active filters