


Security policy (edit with Customer reassurance module)

Delivery policy (edit with Customer reassurance module)

Return policy (edit with Customer reassurance module)
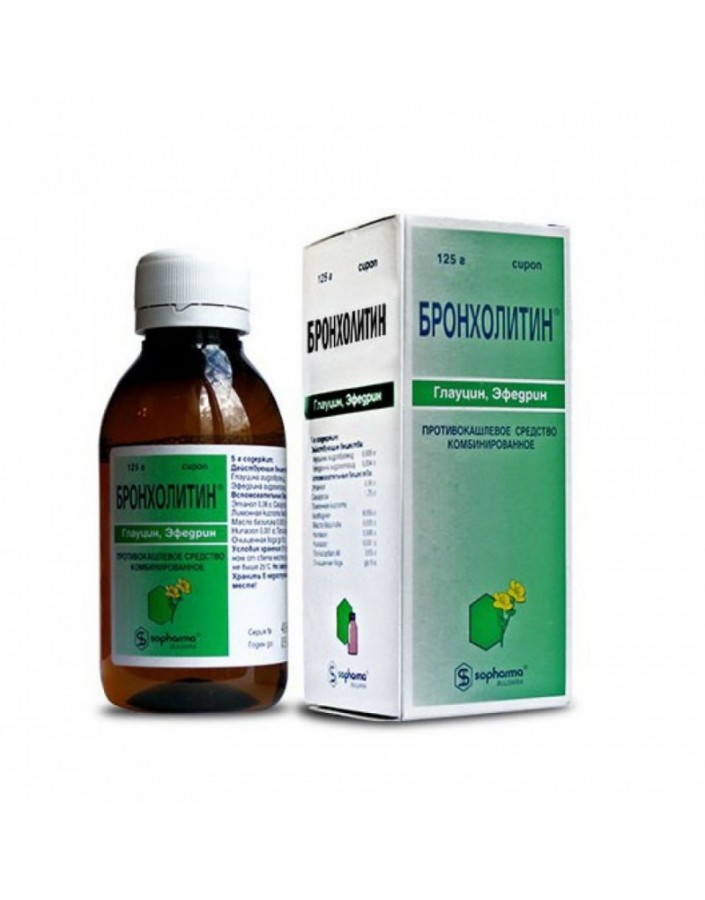
Bronholitin syrup transparent, from light yellow to yellow-green color, with a specific smell of basil oil.
5 ml of syrup contains glaucine hydrobromide 5.75 mg, ephedrine hydrochloride 4.6 mg;
Excipients: citric acid anhydride, basil oil, ethanol 96% (1.7 vol.%), sucrose, sodium methylhydroxybenzoate, propyl parahydroxybenzoate, polysorbate 80, distilled water.
125 g per vial complete with a measuring spoon in a carton box.
Bronholitin - combination drug. It has antitussive and bronchodilatory effect.
Glaucine hydrobromide suppresses the cough center, without leading to respiratory depression, the development of constipation and drug dependence.
Ephedrine expands the bronchi, stimulates breathing, and, due to the vasoconstrictor action, eliminates the swelling of the bronchial mucosa.
Basil oil has a slight sedative, antimicrobial and antispasmodic effect.
Dry cough for various diseases of the respiratory tract, including:
- acute inflammatory diseases of the upper respiratory tract;
- acute and chronic bronchitis;
- tracheobronchitis;
- pneumonia;
- bronchial asthma;
- bronchiectasis;
- whooping cough.
- arterial hypertension;
- CHD;
- severe organic heart disease;
- heart failure;
- pheochromocytoma;
- thyrotoxicosis;
- insomnia;
- glaucoma;
- hyperplasia of the prostate gland with clinical manifestations;
- thyrotoxicosis;
- I trimester of pregnancy;
- lactation (breastfeeding period);
- hypersensitivity to the drug.
For adults designate 10 ml (2 scoops) 3-4 times / day.
Children aged 3 to 10 years - 5 ml (1 scoop) 3 times / day; children over the age of 10 - 10 ml (2 scoops) 3 times / day.
Cardiovascular: tachycardia, extrasystole, increased blood pressure.
From the side of the central nervous system: tremor, agitation, insomnia, dizziness, blurred vision; children have drowsiness.
Gastrointestinal: nausea, vomiting, anorexia, constipation.
From the reproductive system: increased libido, dysmenorrhea.
Urogenital: difficulty urinating, urinary retention in patients with prostatic hyperplasia.
Dermatologic: rash, increased sweating.
Other: tachyphylaxis.
Precautions should appoint Bronholitin patients with a tendency to develop drug dependence.
Bronholitin can be used simultaneously with antibiotics, antipyretics and Vitamins.
When using the drug Bronholitin, it is necessary to take into account that it contains 1.7% by volume of ethanol, each dose contains up to 0.069 g of ethanol. In this regard, Bronholitin should be prescribed with caution to patients with liver diseases, alcoholism, epilepsy, brain diseases, during pregnancy and children.
Influence on ability to drive motor transport and control mechanisms
Due to the possible occurrence of dizziness and visual impairment, caution should be exercised when performing work that requires increased attention and quickness of psychomotor reactions, including when driving a car and working with mechanisms.
Symptoms: nausea, vomiting, loss of appetite, agitation, impaired blood circulation, extremity tremor, dizziness, sweating, difficulty urinating.
Treatment: gastric lavage, symptomatic therapy.
Ephedrine, which is part of the drug, weakens the effect of drugs and hypnotics.
When ephedrine is used simultaneously with cardiac glycosides, quinidine, tricyclic antidepressants, the risk of arrhythmias increases.
With simultaneous use of ephedrine with reserpine and MAO inhibitors, a dramatic increase in blood pressure is possible.
With simultaneous use of ephedrine with non-selective beta-blockers may decrease bronchodilator action.
There is no data on the drug interaction of glaucine with other drugs.
The drug should be stored in a dark place at a temperature not exceeding 25 ° C; do not freeze.
Bronholitin